Observational study of SARS-CoV-2 antibody immune response in a cohort of patients at a North Suburban Chicago, Illinois, in a physician’s practice
Abstract
Background
Introduction
Methods

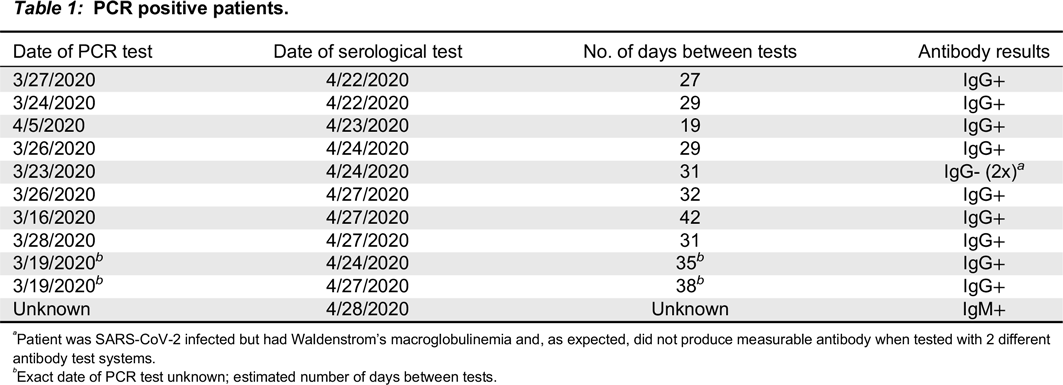
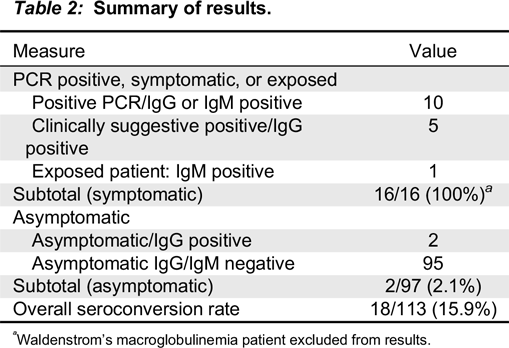
Results
Discussion
Limitations
Conclusion
Disclosures
Funding
Acknowledgements
Footnote
REFERENCES
Information & Authors
Information
Published In

History
Copyright
Authors
Metrics & Citations
Metrics
Other Metrics
Citations
Cite As
Export Citations
If you have the appropriate software installed, you can download article citation data to the citation manager of your choice. Simply select your manager software from the list below and click Download.
Cited by
View Options
View options
Login options
Check if you access through your login credentials or your institution to get full access on this article.


